Selenium
About this schools Wikipedia selection
This Schools selection was originally chosen by SOS Children for schools in the developing world without internet access. It is available as a intranet download. Click here to find out about child sponsorship.
| Selenium | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
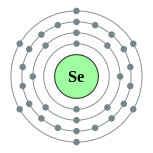
34Se
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
black and red allotropes |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| General properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Name, symbol, number | selenium, Se, 34 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pronunciation | / s ɨ ˈ l iː n i ə m / si-LEE-nee-əm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Element category | nonmetal sometimes considered a metalloid |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Group, period, block | 16 (chalcogens), 4, p | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Standard atomic weight | 78.96 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electron configuration | [Ar] 3d10 4s2 4p4 2, 8, 18, 6 |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| History | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Discovery | Jöns Jakob Berzelius and Johann Gottlieb Gahn (1817) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| First isolation | Jöns Jakob Berzelius and Johann Gottlieb Gahn (1817) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phase | solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density (near r.t.) | (gray) 4.81 g·cm−3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density (near r.t.) | (alpha) 4.39 g·cm−3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density (near r.t.) | (vitreous) 4.28 g·cm−3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Liquid density at m.p. | 3.99 g·cm−3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point | 494 K, 221 °C, 430 °F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Boiling point | 958 K, 685 °C, 1265 °F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Critical point | 1766 K, 27.2 MPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heat of fusion | (gray) 6.69 kJ·mol−1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heat of vaporization | 95.48 kJ·mol−1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molar heat capacity | 25.363 J·mol−1·K−1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Vapor pressure | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Oxidation states | 6, 4, 2, 1, -2 (strongly acidic oxide) |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electronegativity | 2.55 (Pauling scale) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ionization energies | 1st: 941.0 kJ·mol−1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2nd: 2045 kJ·mol−1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3rd: 2973.7 kJ·mol−1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic radius | 120 pm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Covalent radius | 120±4 pm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Van der Waals radius | 190 pm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Miscellanea | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Crystal structure | hexagonal |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Magnetic ordering | diamagnetic | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thermal conductivity | (amorphous) 0.519 W·m−1·K−1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thermal expansion | (25 °C) (amorphous) 37 µm·m−1·K−1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Speed of sound (thin rod) | (20 °C) 3350 m·s−1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Young's modulus | 10 GPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Shear modulus | 3.7 GPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Bulk modulus | 8.3 GPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Poisson ratio | 0.33 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mohs hardness | 2.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Brinell hardness | 736 MPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS registry number | 7782-49-2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Most stable isotopes | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Main article: Isotopes of selenium | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Selenium is a chemical element with symbol Se and atomic number 34. It is a nonmetal with properties that are intermediate between those of its periodic table column-adjacent chalcogen elements sulfur and tellurium. It rarely occurs in its elemental state in nature, or as pure ore compounds. Selenium (Greek σελήνη selene meaning "Moon") was discovered in 1817 by Jöns Jacob Berzelius, who noted the similarity of the new element to the previously-known tellurium (named for the Earth).
Selenium is found impurely in metal sulfide ores, where it partially replaces the sulfur. Commercially, selenium is produced as a byproduct in the refining of these ores, most often during copper production. Minerals that are pure selenide or selenate compounds are known, but are rare. The chief commercial uses for selenium today are in glassmaking and in pigments. Selenium is a semiconductor and is used in photocells. Uses in electronics, once important, have been mostly supplanted by silicon semiconductor devices. Selenium continues to be used in a few types of DC power surge protectors and one type of fluorescent quantum dot.
Selenium salts are toxic in large amounts, but trace amounts are necessary for cellular function in many organisms, including all animals. Selenium is a component of the antioxidant enzymes glutathione peroxidase and thioredoxin reductase (which indirectly reduce certain oxidized molecules in animals and some plants). It is also found in three deiodinase enzymes, which convert one thyroid hormone to another. Selenium requirements in plants differ by species, with some plants requiring relatively large amounts, and others apparently requiring none.
Characteristics
Physical properties
Selenium exists in several allotropes that interconvert upon heating and cooling carried out at different temperatures and rates. As prepared in chemical reactions, selenium is usually amorphous, brick-red powder. When rapidly melted, it forms the black, vitreous form, which is usually sold industrially as beads. The structure of black selenium is irregular and complex and consists of polymeric rings with up to 1000 atoms per ring. Black Se is a brittle, lustrous solid that is slightly soluble in CS2. Upon heating, it softens at 50 °C and converts to gray selenium at 180 °C; the transformation temperature is reduced by presence of halogens and amines.
The red-colored α, β and γ forms are produced from solutions of black selenium by varying evaporation rates of the solvent (usually CS2). They all have relatively low, monoclinic crystal symmetries and contain nearly identical puckered Se8 rings arranged in different fashions, as in sulfur. The packing is most dense in the α form. In the Se8 rings, the Se-Se distance is 233.5 pm and Se-Se-Se angle is 105.7 degrees. Other selenium allotropes may contain Se6 or Se7 rings.
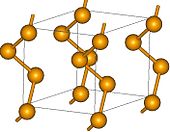
The most stable and dense form of selenium has a gray colour and hexagonal crystal lattice consisting of helical polymeric chains, wherein the Se-Se distance is 237.3 pm and Se-Se-Se angle is 130.1 degrees. The minimum distance between chains is 343.6 pm. Gray Se is formed by mild heating of other allotropes, by slow cooling of molten Se, or by condensing Se vapors just below the melting point. Whereas other Se forms are insulators, gray Se is a semiconductor showing appreciable photoconductivity. Contrary to other allotropes, it is insoluble in CS2. It resists oxidation by air and is not attacked by non-oxidizing acids. With strong reducing agents, it forms polyselenides. Selenium does not exhibit the unusual changes in viscosity that sulfur undergoes when gradually heated.
Isotopes
Selenium has six naturally occurring isotopes, five of which are stable: 74Se, 76Se, 77Se, 78Se, and 80Se. The last three also occur as fission products, along with 79Se, which has a half-life of 327,000 years. The final naturally occurring isotope, 82Se, has a very long half-life (~1020 yr, decaying via double beta decay to 82Kr), which, for practical purposes, can be considered to be stable. Twenty-three other unstable isotopes have been characterized.
See also Selenium-79 for more information on recent changes in the measured half-life of this long-lived fission product, important for the dose calculations performed in the frame of the geological disposal of long-lived radioactive waste.
Chemical compounds
Selenium compounds commonly exist in the oxidation states −2, +2, +4, and +6.
Chalcogen compounds
Selenium forms two oxides: selenium dioxide (SeO2) and selenium trioxide (SeO3). Selenium dioxide is formed by the reaction of elemental selenium with oxygen:
- Se8 + 8 O2 → 8 SeO2
It is a polymeric solid that forms monomeric SeO2 molecules in the gas phase. It dissolves in water to form selenous acid, H2SeO3. Selenous acid can also be made directly by oxidizing elemental selenium with nitric acid:
- 3 Se + 4 HNO3 + H2O → 3 H2SeO3 + 4 NO
Unlike sulfur, which forms a stable trioxide, selenium trioxide is thermodynamically unstable and decomposes to the dioxide above 185 °C:
- 2 SeO3 → 2 SeO2 + O2 (ΔH = −54 kJ/mol)
Selenium trioxide is produced in the laboratory by the reaction of anhydrous potassium selenate (K2SeO4) and sulfur trioxide (SO3).
Salts of selenous acid are called selenites. These include silver selenite (Ag2SeO3) and sodium selenite (Na2SeO3).
Hydrogen sulfide reacts with aqueous selenous acid to produce selenium disulfide:
- H2SeO3 + 2 H2S → SeS2 + 3 H2O
Selenium disulfide consists of 8-membered rings of a nearly statistical distribution of sulfur and selenium atoms. It has an approximate composition of SeS2, with individual rings varying in composition, such as Se4S4 and Se2S6. Selenium disulfide has been use in shampoo as an anti- dandruff agent, an inhibitor in polymer chemistry, a glass dye, and a reducing agent in fireworks.
Selenium trioxide may be synthesized by dehydrating selenic acid, H2SeO4, which is itself produced by the oxidation of selenium dioxide with hydrogen peroxide:
- SeO2 + H2O2 → H2SeO4
Hot, concentrated selenic acid is capable of dissolving gold, forming gold(III) selenate.
Halogen compounds
Iodides of selenium are not well known. The only stable chloride is selenium monochloride (Se2Cl2), which might be better known as selenium(I) chloride; the corresponding bromide is also known. These species are structurally analogous to the corresponding disulfur dichloride. Selenium dichloride is an important reagent in the preparation of selenium compounds (e.g. the preparation of Se7). It is prepared by treating selenium with sulfuryl chloride (SO2Cl2). Selenium reacts with fluorine to form selenium hexafluoride:
- Se8 + 24 F2 → 8 SeF6
In comparison with its sulfur counterpart ( sulfur hexafluoride), selenium hexafluoride (SeF6) is more reactive and is a toxic pulmonary irritant. Some of the selenium oxyhalides, such as selenium oxyfluoride (SeOF2) and selenium oxychloride (SeOCl2) have been used as specialty solvents.
Selenides
Analogous to the behaviour of other chalcogens, selenium forms a dihydride H2Se. It is a strongly odiferous, toxic, and colorless gas. It is more acidic than H2S. In solution it ionizes to HSe-. The selenide dianion Se2- forms a variety of compounds, including the minerals from which selenium is obtained commercially. Illustrative selenides include mercury selenide (HgSe), lead selenide (PbSe), zinc selenide (ZnSe), and copper indium gallium diselenide (Cu(Ga,In)Se2). These materials are semiconductors. With highly electropositive metals, such as aluminium, these selenides are prone to hydrolysis:
- Al2Se3 + 6 H2O → Al2O3 + 6 H2Se
Alkali metal selenides react with selenium to form polyselenides, Se2−
x, which exist as chains.
Other compounds
Tetraselenium tetranitride, Se4N4, is an explosive orange compound analogous to tetrasulfur tetranitride (S4N4). It can be synthesized by the reaction of selenium tetrachloride (SeCl4) with [((CH3)3Si)2N]2Se.
Selenium reacts with cyanides to yield selenocyanates:
- 8 KCN + Se8 → 8 KSeCN
Organoselenium compounds
Selenium, especially in the II oxidation state, forms stable bonds to carbon, which are structurally analogous to the corresponding organosulfur compounds. Especially common are selenides (R2Se, analogues of thioethers), diselenides (R2Se2, analogues of disulfides), and selenols (RSeH, analogues of thiols). Representatives of selenides, diselenides, and selenols include respectively selenomethionine, diphenyldiselenide, and benzeneselenol. The sulfoxide in sulfur chemistry is represented in selenium chemistry by the selenoxides (formula RSe(O)R), which are intermediates in organic synthesis, as illustrated by the selenoxide elimination reaction. Consistent with trends indicated by the double bond rule, selenoketones, R(C=Se)R, and selenaldehydes, R(C=Se)H, are rarely observed.
History
Selenium (Greek σελήνη selene meaning "Moon") was discovered in 1817 by Jöns Jakob Berzelius and Johan Gottlieb Gahn. Both chemists owned a chemistry plant near Gripsholm, Sweden producing sulfuric acid by the lead chamber process. The pyrite from the Falun mine created a red precipitate in the lead chambers which was presumed to be an arsenic compound, and so the pyrite's use to make acid was discontinued. Berzelius and Gahn wanted to use the pyrite and they also observed that the red precipitate gave off a smell like horseradish when burned. This smell was not typical of arsenic, but a similar odour was known from tellurium compounds. Hence, Berzelius's first letter to Alexander Marcet stated that this was a tellurium compound. However, the lack of tellurium compounds in the Falun mine minerals eventually led Berzelius to reanalyze the red precipitate, and in 1818 he wrote a second letter to Marcet describing a newly found element similar to sulfur and tellurium. Because of its similarity to tellurium, named for the Earth, Berzelius named the new element after the Moon.
In 1873, Willoughby Smith found that the electrical resistance of grey selenium was dependent on the ambient light. This led to its use as a cell for sensing light. The first of commercial products using selenium were developed by Werner Siemens in the mid-1870s. The selenium cell was used in the photophone developed by Alexander Graham Bell in 1879. Selenium transmits an electric current proportional to the amount of light falling on its surface. This phenomenon was used in the design of light meters and similar devices. Selenium's semiconductor properties found numerous other applications in electronics. The development of selenium rectifiers began during the early 1930s, and these replaced copper oxide rectifiers because of their superior efficiencies. These lasted in commercial applications until the 1970s, following which they were replaced with less expensive and even more efficient silicon rectifiers.
Selenium came to medical notice later because of its toxicity to human beings working in industries. Selenium was also recognized as an important veterinary toxin, which is seen in animals that have eaten high-selenium plants. In 1954, the first hints of specific biological functions of selenium were discovered in microorganisms. Its essentiality for mammalian life was discovered in 1957. In the 1970s, it was shown to be present in two independent sets of enzymes. This was followed by the discovery of selenocysteine in proteins. During the 1980s, it was shown that selenocysteine is encoded by the codon UGA. The recoding mechanism was worked out first in bacteria and then in mammals (see SECIS element).
Occurrence
Native (i.e., elemental) selenium is a rare mineral, which does not usually form good crystals, but, when it does, they are steep rhombohedra or tiny acicular (hair-like) crystals. Isolation of selenium is often complicated by the presence of other compounds and elements.
Selenium occurs naturally in a number of inorganic forms, including selenide-, selenate-, and selenite-containing minerals, but these minerals are rare. The common mineral selenite is not a selenium mineral, and contains no selenite ion, but is rather a type of gypsum (calcium sulfate hydrate) named like selenium for the moon well before the discovery of selenium. Selenium is most commonly found quite impurely, replacing a small part of the sulfur in sulfide ores of many metals.
In living systems, selenium is found in the amino acids selenomethionine, selenocysteine, and methylselenocysteine. In these compounds, selenium plays a role analogous to that of sulfur. Another naturally occurring organoselenium compound is dimethyl selenide.
Certain solids are selenium-rich, and selenium can be bioconcentrated by certain plants. In soils, selenium most often occurs in soluble forms such as selenate (analogous to sulfate), which are leached into rivers very easily by runoff. Ocean water contains significant amounts of selenium.
Anthropogenic sources of selenium include coal burning and the mining and smelting of sulfide ores.
Production
Selenium is most commonly produced from selenide in many sulfide ores, such as those of copper, silver, or lead. Electrolytic metal refining is particularly conducive to producing selenium as a byproduct, and it is obtained from the anode mud of copper refineries. Another source was the mud from the lead chambers of sulfuric acid plants but this method to produce sulfuric acid is no longer used. These muds can be processed by a number of means to obtain selenium. However, most elemental selenium comes as a byproduct of refining copper or producing sulfuric acid. Since the invention of solvent extraction and electrowinning (SX/EW) for the production of copper this method takes an increasing share of the world wide copper production. This changes the availability of selenium because only a comparably small part of the selenium in the ore is leached together with the copper.
Industrial production of selenium usually involves the extraction of selenium dioxide from residues obtained during the purification of copper. Common production from the residue then begins by oxidation with sodium carbonate to produce selenium dioxide. The selenium dioxide is then mixed with water and the solution is acidified to form selenous acid ( oxidation step). Selenous acid is bubbled with sulfur dioxide ( reduction step) to give elemental selenium.
About 2,000 tonnes of selenium has been produced in 2011 worldwide, mostly in Germany (650 t), Japan (630 t), Belgium (200 t) and Russia (140 t), and the total reserves were estimated at 93,000 tonnes. These data however exclude two major producers, the United States and China. The price has been relatively stable during 2004–2010 at ~30 US dollars per pound (per 100-pound lot) but has increased to 65 $/lb in 2011. A previous sharp increase was observed in 2004 from 4–5 to 27 $/lb. The consumption in 2010 was divided as follows: metallurgy – 30%, glass manufacturing – 30%, agriculture – 10%, chemicals and pigments – 10%, electronics – 10%. China is the dominant consumer of selenium at 1,500–2,000 tonnes/year.
Applications
Manganese electrolysis
During the electro winning of manganese an addition of selenium dioxide decreases the power necessary to operate the electrolysis cells. China is the largest consumer of selenium dioxide for this purpose. For every tonne of manganese an average of 2 kg selenium oxide is used.
Glass production
The largest commercial use of Se, accounting for about 50% of consumption, is for the production of glass. Se compounds confer a red color to glass. This color cancels out the green or yellow tints that arise from iron impurities that are typical for most glass. For this purpose various selenite and selenate salts are added. For other applications, the red colour may be desirable, in which case mixtures of CdSe and CdS are added.
Alloys
Selenium is used with bismuth in brasses to replace more toxic lead. The regulation of lead in drinking water applications with the Safe Drinking Water Act of 1974 made a reduction of lead in brass necessary. The new brass is marketed under the name EnviroBrass. Like lead and sulfur, selenium improves the machinability of steel at concentrations of 0.15%. The same improvement is also observed in copper alloys and therefore selenium is also used in machinable copper alloys.
Solar cells
Copper indium gallium selenide is a material used in the production of solar cells.
Other uses
Small amounts of organoselenium compounds are used to modify the vulcanization catalysts used in the production of rubber.
The demand for selenium by the electronics industry is declining, despite a number of continuing applications. Because of its photovoltaic and photoconductive properties, selenium is used in photocopying, photocells, light meters and solar cells. Its use as a photoconductor in plain-paper copiers once was a leading application but in the 1980s, the photoconductor application declined (although it was still a large end-use) as more and more copiers switched to the use of organic photoconductors. It was once widely used in selenium rectifiers. These uses have mostly been replaced by silicon-based devices or are in the process of being replaced. The most notable exception is in power DC surge protection, where the superior energy capabilities of selenium suppressors make them more desirable than metal oxide varistors.
Zinc selenide was the first material for blue LEDs but gallium nitride is dominating the market now. Cadmium selenide has recently played an important part in the fabrication of quantum dots. Sheets of amorphous selenium convert x-ray images to patterns of charge in xeroradiography and in solid-state, flat-panel x-ray cameras.
Selenium is a catalyst in some chemical reactions but it is not widely used because of issues with toxicity. In X-ray crystallography, incorporation of one or more selenium atoms in place of sulfur helps with Multi-wavelength anomalous dispersion and Single wavelength anomalous dispersion phasing.
Selenium is used in the toning of photographic prints, and it is sold as a toner by numerous photographic manufacturers. Its use intensifies and extends the tonal range of black-and-white photographic images and improves the permanence of prints.
75Se is used as a gamma source in industrial radiography.
Biological role
Although it is toxic in large doses, selenium is an essential micronutrient for animals. In plants, it occurs as a bystander mineral, sometimes in toxic proportions in forage (some plants may accumulate selenium as a defense against being eaten by animals, but other plants such as locoweed require selenium, and their growth indicates the presence of selenium in soil). See more on plant nutrition below.
Selenium is a component of the unusual amino acids selenocysteine and selenomethionine. In humans, selenium is a trace element nutrient that functions as cofactor for reduction of antioxidant enzymes, such as glutathione peroxidases and certain forms of thioredoxin reductase found in animals and some plants (this enzyme occurs in all living organisms, but not all forms of it in plants require selenium).
The glutathione peroxidase family of enzymes (GSH-Px) catalyze certain reactions that remove reactive oxygen species such as hydrogen peroxide and organic hydroperoxides:
- 2 GSH + H2O2----GSH-Px → GSSG + 2 H2O
Selenium also plays a role in the functioning of the thyroid gland and in every cell that uses thyroid hormone, by participating as a cofactor for the three of the four known types of thyroid hormone deiodinases, which activate and then deactivate various thyroid hormones and their metabolites: the iodothyronine deiodinases are the subfamily of deiodinase enzymes that use selenium as the otherwise rare amino acid selenocysteine. (Only the deiodinase iodotyrosine deiodinase, which works on the last break-down products of thyroid hormone, does not use selenium).
Selenium may inhibit Hashimoto's disease, in which the body's own thyroid cells are attacked as alien. A reduction of 21% on TPO antibodies was reported with the dietary intake of 0.2 mg of selenium.
Increased dietary selenium intakes reduce the effects of mercury toxicity and it is now recognized that the molecular mechanism of mercury toxicity involves irreversible inhibition of selenoenzymes that are required to prevent and reverse oxidative damage in brain and endocrine tissues.
Evolution in biology
From about three billion years ago, prokaryotic selenoprotein families drive the evolution of selenocysteine. Selenium is incorporated into several prokaryotic selenoprotein families in bacteria, archaea and eukaryotes as selenocysteine, where selenoprotein peroxiredoxins protect bacterial and eukaryotic cells against oxidative damage. Selenoprotein families of GSH-Px and the deiodinases of eukaryotic cells seem to have a bacterial phylogenetic origin. The selenocysteine-containing form occurs in species as diverse as green algae, diatoms, sea urchin, fish and chicken. Selenium enzymes are involved in utilization of the small reducing molecules glutathione and thioredoxin. One family of selenium-containing molecules (the glutathione peroxidases) destroy peroxide and repair damaged peroxidized cell membranes, using glutathione. Another selenium-containing enzyme in some plants and in animals ( thioredoxin reductase) generates reduced thioredoxin, a dithiol that serves as an electron source for peroxidases and also the important reducing enzyme ribonucleotide reductase that makes DNA precursors from RNA precursors.
Trace elements involved in GSH-Px and superoxide dismutase enzymes activities, i.e. selenium, vanadium, magnesium, copper, and zinc, may have been lacking in some terrestrial mineral-deficient areas. Marine organisms retained and sometimes expanded their seleno-proteomes, whereas the seleno-proteomes of some terrestrial organisms were reduced or completely lost. These findings suggest that, with the exception of vertebrates, aquatic life supports selenium utilization, whereas terrestrial habitats lead to reduced use of this trace element. Marine fishes and vertebrate thyroid glands have the highest concentration of selenium and iodine. From about 500 Mya, freshwater and terrestrial plants slowly optimized the production of "new" endogenous antioxidants such as ascorbic acid (Vitamin C), polyphenols (including flavonoids), tocopherols, etc. A few of these appeared more recently, in the last 50–200 million years, in fruits and flowers of angiosperm plants. In fact, the angiosperms (the dominant type of plant today) and most of their antioxidant pigments evolved during the late Jurassic period.
The deiodinase isoenzymes constitute another family of eukaryotic selenoproteins with identified enzyme function. Deiodinases are able to extract electrons from iodides, and iodides from iodothyronines. They are, thus, involved in thyroid-hormone regulation, participating in the protection of thyrocytes from damage by H2O2 produced for thyroid-hormone biosynthesis. About 200 Mya, new selenoproteins were developed as mammalian GSH-Px enzymes.
Indicator plant species
Certain species of plants are considered indicators of high selenium content of the soil, since they require high levels of selenium to thrive. The main selenium indicator plants are Astragalus species (including some locoweeds), prince's plume ( Stanleya sp.), woody asters ( Xylorhiza sp.), and false goldenweed ( Oonopsis sp.)
Medical use
The substance loosely called selenium sulfide (approximate formula SeS2) is the active ingredient in some anti-dandruff shampoos. The selenium compound kills the scalp fungus Malassezia, which causes shedding of dry skin fragments. The ingredient is also used in body lotions to treat Tinea versicolor due to infection by a different species of Malassezia fungus.
Detection in biological fluids
Selenium may be measured in blood, plasma, serum or urine to monitor excessive environmental or occupational exposure, confirm a diagnosis of poisoning in hospitalized victims or to assist in a forensic investigation in a case of fatal overdosage. Some analytical techniques are capable of distinguishing organic from inorganic forms of the element. Both organic and inorganic forms of selenium are largely converted to monosaccharide conjugates (selenosugars) in the body prior to being eliminated in the urine. Cancer patients receiving daily oral doses of selenothionine may achieve very high plasma and urine selenium concentrations.
Toxicity
Although selenium is an essential trace element, it is toxic if taken in excess. Exceeding the Tolerable Upper Intake Level of 400 micrograms per day can lead to selenosis. This 400 microgram ( µg) Tolerable Upper Intake Level is based primarily on a 1986 study of five Chinese patients who exhibited overt signs of selenosis and a follow up study on the same five people in 1992. The 1992 study actually found the maximum safe dietary Se intake to be approximately 800 micrograms per day (15 micrograms per kilogram body weight), but suggested 400 micrograms per day to not only avoid toxicity, but also to avoid creating an imbalance of nutrients in the diet and to account for data from other countries. In China, people who ingested corn grown in extremely selenium-rich stony coal (carbonaceous shale) have suffered from selenium toxicity. This coal was shown to have selenium content as high as 9.1%, the highest concentration in coal ever recorded in literature.
Symptoms of selenosis include a garlic odour on the breath, gastrointestinal disorders, hair loss, sloughing of nails, fatigue, irritability, and neurological damage. Extreme cases of selenosis can result in cirrhosis of the liver, pulmonary edema, and death. Elemental selenium and most metallic selenides have relatively low toxicities because of their low bioavailability. By contrast, selenates and selenites are very toxic, having an oxidant mode of action similar to that of arsenic trioxide. The chronic toxic dose of selenite for humans is about 2400 to 3000 micrograms of selenium per day for a long time. Hydrogen selenide is an extremely toxic, corrosive gas. Selenium also occurs in organic compounds, such as dimethyl selenide, selenomethionine, selenocysteine and methylselenocysteine, all of which have high bioavailability and are toxic in large doses.
On 19 April 2009, 21 polo ponies died shortly before a match in the United States Polo Open. Three days later, a pharmacy released a statement explaining that the horses had received an incorrect dose of one of the ingredients used in a vitamin/mineral supplement compound that had been incorrectly compounded by a compounding pharmacy. Analysis of blood levels of inorganic compounds in the supplement indicated the selenium concentrations were ten to fifteen times higher than normal in the horses' blood samples, and 15 to 20 times higher than normal in their liver samples. It was later confirmed that selenium was the ingredient in question.
Selenium poisoning of water systems may result whenever new agricultural runoff courses through normally dry, undeveloped lands. This process leaches natural soluble selenium compounds (such as selenates) into the water, which may then be concentrated in new "wetlands" as the water evaporates. High selenium levels produced in this fashion have been found to have caused certain congenital disorders in wetland birds.
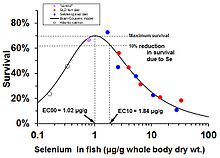
In fish and other wildlife, low levels of selenium cause deficiency while high levels cause toxicity. For example, in salmon, the optimal concentration of selenium in the fish tissue (whole body) is about 1 microgram selenium per gram of tissue (dry weight). At levels much below that concentration, young salmon die from selenium deficiency; much above that level they die from toxic excess.
Deficiency
Selenium deficiency is rare in healthy, well-nourished individuals. It can occur in patients with severely compromised intestinal function, those undergoing total parenteral nutrition, and in those of advanced age (over 90). Also, people dependent on food grown from selenium-deficient soil are at risk. Although New Zealand has low levels of selenium in its soil, adverse health effects have not been detected.
Selenium deficiency as defined by low (<60% of normal) selenoenzyme activity levels in brain and endocrine tissues only occurs when a low selenium status is linked with an additional stress, such as high exposures to mercury or as a result of increased oxidant stress due to vitamin E deficiency.
There are interactions between selenium and other nutrients, such as iodine and vitamin E. The effect of selenium deficiency on health remains uncertain, particularly in relation to Kashin-Beck disease.
In some regions (e.g. various regions within North America) where low available selenium levels in soil lead to low concentrations in dry matter of plants, Se deficiency in some animal species may occur unless dietary (or injected) selenium supplementation is done. Ruminants are particularly susceptible. In general, absorption of dietary selenium is lower in ruminants than in non-ruminants, and is lower from forages than from grain. Ruminants grazing certain forages, e.g. some white clover varieties containing cyanogenic glycosides, may have higher selenium requirements, presumably because of cyanide from the aglycone released by glucosidase activity in the rumen and inactivation of glutathione peroxidases due to absorbed cyanide's effect on the glutathione moiety. Neonate ruminants at risk of WMD (white muscle disease) may be administered both selenium and vitamin E by injection; some of the WMD myopathies respond only to selenium, some only to vitamin E, and some to either.
Controversial health effects
A number of correlative epidemiological studies have implicated selenium deficiency (as measured by blood levels) in a number of serious or chronic diseases, such as cancer, diabetes, HIV/AIDS, and tuberculosis. In addition, selenium supplementation has been found to be a chemopreventive for some types of cancer in some types of rodents. However, in randomized, blinded, controlled prospective trials in humans, selenium supplementation has not succeeded in reducing the incidence of any disease, nor has a meta-analysis of such selenium supplementation studies detected a decrease in overall mortality.